© Getty Images/selvanegra
Graphic representation of COVID-19 binding to the ACE2 receptor on human cells via its spike protein (red)
I’ve been reading case reports of heart damage in people infected with COVID-19. In fact, someone in my close circle had an episode of fulminant myopericarditis and a strange lung pathology last December, from which he miraculously recovered. I deem SARS-CoV-2 (the virus that causes the disease COVID-19) a likely suspect behind my friend’s illness, even though this occurred a few months before the pandemic officially began in Europe.
The following LiveScience article provides some clues as to how both the
heart and lungs can be seriously affected during a COVID-19 infection:
The mysterious connection between the coronavirus and the heart
“We’re seeing cases of people who don’t have an underlyingheart disease, who are getting heart damage,” said Dr. Erin Michos, the associate director of preventive cardiology at Johns Hopkins School of Medicine. Heart damage isn’t typical in mild cases of COVID-19, and tends to occur more often in patients who have severe symptoms and are hospitalized, she said. […]
Both heart cells and lung cells are covered with surface proteins known as angiotensin-converting enzyme 2 (ACE2) — these molecules serve as “doorways” for the virus to enter cells.But this enzyme is a “double-edged sword,” she said. On one hand, theACE2molecule acts as a gateway for the virus to enter the cell and replicate, but on the other hand, it normally serves a “protective” function, Michos said.
When tissues in the body are damaged — either by an invading virus such as SARS-CoV-2 or by other means, the body’s natural healing response involves releasing inflammatory molecules, such as small proteins called cytokines, into the bloodstream. But paradoxically, too much inflammation can actually make things worse. The ACE2 enzyme acts as an anti-inflammatory, keeping immune cells from inflicting more damage on the body’s own cells.
But when the virus latches onto ACE2 proteins, these proteins get knocked out of commission, possibly reducing the anti-inflammatory protection that they give. So the virus may be acting as a double-whammy by damaging cells directly and preventing the body from protecting tissues from inflammatory damage.
“If the heart muscle is inflamed and damaged by the virus, the heart can’t function,” she said.
The novel coronavirus might also indirectly damage the heart. In this scenario, the patient’s immune system winds up “going haywire,” Michos said. This scenario has played out in some really sick patients who have highly elevated inflammatory markers — or proteins that signal high levels of inflammation in the body.
This is called a “cytokine storm,” Michos said. Cytokine storms damage organs throughout the body, including the heart and liver, she added. It’s not clear why some people have such an elevated response compared with others, but some people could be genetically prone to it, she added. […]
It’s really hard to tease out whether having more ACE2 is helpful or harmful, as these proteins are how the virus enters the cells, but also known to protect the cells against injury, Michos said.
A pre-print on smoking and hospitalization for COVID-19
brings up this same issue of whether the ACE2 molecule gateway is
harmful or beneficial. We all know how smoking is terrible for people
and is related to many illnesses, right? Well, the puzzling thing is
that smokers and ex-smokers seem to fare better than non-smokers
when it comes to COVID-19 complications. The following paper “raises
the hypothesis that nicotine may have beneficial effects on COVID-19″:
Smoking, vaping and hospitalization for COVID-19
Konstantinos Farsalinos1, Anastasia Barbouni1, Raymond Niaura2
- University of West Attica, Athens, Greece
- New York University, United States
Abstract
The study purpose was to examine the prevalence of current e-cigarette use and current smoking among hospitalized patients with COVID-19 in China, considering the high population smoking prevalence in the country (26.6%, 50.5% in males and 2.1% in females). A systematic research of the literature (PubMed) was performed on April 1. Out of 432 studies, we identified 13 studies examining the clinical characteristics of a total of 5960 hospitalized COVID-19 patients that presented data on the smoking status. No study reported e-cigarette use among COVID-19 patients. The prevalence of current smoking ranged from 1.4% to 12.6%. The random effect pooled prevalence of current smoking was 6.5% (95%CI: 4.9-8.2%). This preliminary analysis does not support the argument that current smoking is a risk factor for hospitalization for COVID-19. Instead, these consistent observations, which are further emphasized by the low prevalence of current smoking among COVID-19 patients in the US (1.3%), raises the hypothesis that nicotine may have beneficial effects on COVID-19.This could be attributed to its immunomodulatory effects and its interaction with the renin-angiotensin system. However, other confounding factors need to be considered and the accuracy of the recorded smoking status needs to be determined. However, the results were remarkably consistent across all studies and were recently verified in the first case series of COVID-19 cases in the US. In conclusion, the generalized advice to quit smoking as a measure to improve health risk remains valid, but no recommendation can currently be made concerning the effects of smoking on the risk of hospitalization for COVID-19. No studies recording e-cigarette use status among hospitalized COVID-19 patients were identified. Thus, no recommendation can be made for e-cigarette users. The above-mentioned observations, together with the potential mechanisms through which nicotine interacts with the inflammatory process and the renin-angiotensin-aldosterone axis involved in the development of COVID-19, warrant an urgent investigation of the clinical effects of pharmaceutical nicotine on COVID-19 susceptibility, progression and severity.
To explain why smokers seemed to fare better, the authors speculate that nicotine might have beneficial immunomodulatory effects. However, they further state that interactions between nicotine and the renin-angiotensin-aldosterone axis remain unclear.
At the other end of this debate, the following paper published in The American Journal of Physiology considers nicotine to have a detrimental effect on the body through the same system:
Nicotine and the renin-angiotensin system
doi.org/10.1152/ajpregu.00099.2018
This review focuses on the interaction between nicotine and the renin-angiotensin system (RAS), one of the most important regulatory systems on autonomic, cardiovascular, and pulmonary functions in both health and disease. The literature presented in this review strongly suggests that nicotine alters the homeostasis of the RAS by upregulating the detrimental angiotensin-converting enzyme (ACE)/angiotensin (ANG)-II/ANG II type 1 receptor axis and downregulating the compensatory ACE2/ANG-(1-7)/Mas receptor axis, contributing to the development of CVPD.
This paper included a graph to illustrate what is known about nicotine and angiotensin pathway:
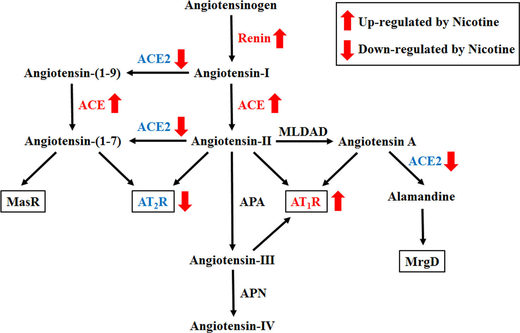
© 2018 the American Physiological Society
Overview of the renin-angiotensin system (RAS) and the interaction between nicotine and the RAS. ACE, angiotensin-converting enzyme; APA, aminopeptidase A; APN, aminopeptidase N; AT1R, angiotensin II type 1 receptor; AT2R, angiotensin II type 2 receptor; MasR, Mas receptor; MLDAD, mononuclear leukocyte-derived aspartate decarboxylase.
Note that, while the authors portray the renin-angiotensin system as one of the most important regulatory systems on autonomic, cardiovascular, and pulmonary functions in both health and disease, they overlook something fundamental. Arguably the most important regulatory system in the body is the parasympathetic branch of the autonomous nervous system, which serves as the body’s master anti-inflammatory automated circuit.
The vagus nerve is the heart of the parasympathetic nervous system. It
is the body’s own ‘anti-stress system’. The vagus nerve is your calming
and relaxing nerve, which extends from your brain down to your abdomen,
dividing into multiple branches that reach your throat, your heart,
lungs, and all your viscera. It is also called ‘the wanderer’, as it
wanders throughout your body. When you take deep breaths,
this wandering nerve is activated and it releases acetylcholine, which
reduces the production of inflammatory molecules like cytokines. Recall
the ‘cytokine storm’ that occurs in severe COVID-19 cases.
Receptors for acetylcholine, also known as cholinergic receptors, fall into two categories based on chemicals that mimic or antagonize the action of acetylcholine on its target cells. In classic studies, nicotine, isolated from tobacco, was one of the chemicals used to distinguish acetylcholine receptors. This is because our bodies contain nicotinic receptors for acetylcholine. People who smoke often undergo slight temporary impairment of cognitive abilities when they quit smoking, until their bodies re-adjust to the cessation of the nicotine influx. This worsening is because nicotine acts as an agonist to mimic acetylcholine receptors, which is important for learning, memory, and cognitive functions.
The production of cytokines, as well as tumor necrosis factor (TNF) and
interleukin-6 (IL-6), protect us against pathogens and promote tissue
repair, but excessive release of cytokines can result in systemic
inflammation, organ failure, and ultimately death, as is happening in
some patients with COVID-19.
Autonomous regulation of local and systemic inflammation – that is, the
molecular link between the brain and the immune system – is done through
the anti-inflammatory cholinergic pathway. This is the
mechanism comprising the vagus nerve and its neurotransmitter
acetylcholine, a process that depends on the alpha-7 sub-unit of the
nicotinic acetylcholine receptor (α7 nAChR).
Molecular model of the α7 nicotinic receptor, thought to be critical for memory, working memory, learning, and attention.
Acetylcholine has a direct inhibitory effect on the production of pro-inflammatory cytokines, thus avoiding the damaging effects from excess production of cytokines. Nicotine, the prototypical acetylcholine nicotinic receptor agonist, attenuates the release of cytokines such as TNF. Vagus nerve stimulation decreases inflammation without unwanted side effects on organ functions such as respiration, heart-rate, and intestinal motility.
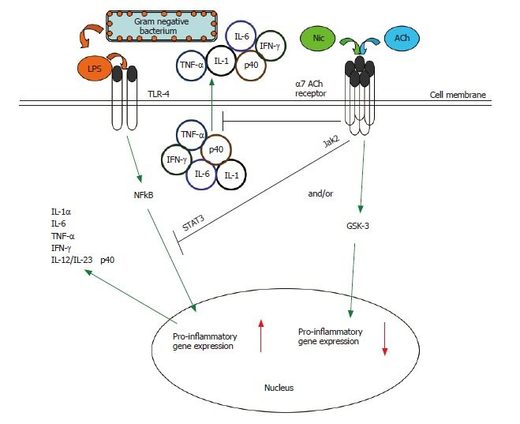
The following graph, where a bacterial infection promotes excessive
cytokine production, gives a summary of the cholinergic
anti-inflammatory pathway, which is activated endogenously (internally)
by acetylcholine (Ach) or exogenously (externally) by nicotine (Nic):
© World J Gastroenterol. 2006 Dec 14; 12(46): 7451–7459.
The α7 nAChR-dependent cholinergic anti-inflammatory pathway, triggered endogenously by acetylcholine or exogenously by nicotine, can suppress the production of several pro-inflammatory cytokines
Nicotine and acetylcholine use the same receptor, the alpha-7 subunit of the nicotinic acetylcholine receptor (α7 nAChR). This receptor – the keyhole for activating this anti-inflammatory response – is also critical for memory, working memory, learning, and attention. That’s why there’s a whole pharmaceutical field looking into this mechanism for possible development and commercialization of non-tobacco-derived nicotinic agonists as anti-inflammatory drugs.
How then can the α7 nAChR-dependent cholinergic anti-inflammatory pathway be so easily overlooked or ignored?
As a practitioner of neural therapy, it’s impossible for me to miss this connection. In fact, all neural therapists know that the autonomous nervous system (or vegetative nervous system) is the most under-appreciated system in mainstream medicine, yet it is central to everything about us: how fast we heal from injuries, how pain is perceived, how well the digestive system is working, how fast the heart beats, how well we regulate stress and inflammation, etc. It connects every system in the body, and data shows that it can do so in non-linear ways.
Your autonomous nervous system is key to regulating many if not most
systems in your body, and it functions effectively at doing so right up
until whichever affliction becomes the ‘straw that breaks the camel’s
back’. However banal some new illness or complaint at first appears, if
it comes along after a slew of prior assaults to your system, chances
are that the autonomous nervous system has ‘collapsed’ and no longer
regulates the chaos in your body.
Your otherwise mundane new illness might – to an allopathic practitioner
unable to identify it via standard tests like endoscopy, ultrasound,
scans, etc. – seem unrelated to systemic or chronic issues. But that’s
likely because he or she is missing the ‘bigger picture’. Neural
therapy, also known as Regulatory Medicine, is based on activating the
parasympathetic ‘anti-stress’ system and its cholinergic
anti-inflammatory pathway so that a person can better regulate stress
and disease.
The autonomic nervous system is a major network of nerve fibers running
from the brain throughout the entire body AND vice versa, with neurons
reaching most of our body’s organs. These neurons release chemicals
which affect target cells in our organs. What this means is that
regardless of the chemicals affecting target cells such as the
renin-angiotensin system, which is a hot topic in the latest
publications of several peer-reviewed journals, the autonomous nervous
system remains the ‘mastermind’ which regulates processes such as
vasoconstriction or vasodilation (constriction or dilation of blood
vessels).
The autonomic nervous system regulates all the involuntary functions in
our body – e.g. the eyes, glands (salivary, lacrimal, sweat), blood
vessels, heart, larynx, trachea, the bronchi, the lungs, the stomach,
the adrenal glands, the kidneys, the pancreas, the intestine, the
bladder and the external genitalia). It is divided into two seemingly
opposing subsystems: the sympathetic and the parasympathetic. The
sympathetic system is associated with a fight-or-flight response to
danger such as an infection. On the contrary, the parasympathetic
system, as we reviewed above, is associated with growth, restoration and
regeneration.
This is why activating the parasympathetic branch of the autonomous
nervous system helps us reduce inflammation run amok. It puts a brake on
the hyperactivity of the sympathetic branch in response to too much
stress from a viral infection.
Let’s go back to the main entry point into cells for COVID-19, the angiotensin-converting enzyme 2 (ACE2):
Angiotensin converting enzyme 2 (ACE2) is known to be present in most organs. ACE2 is attached to the cell membrane of mainly lung alveolar epithelial cells, enterocytes of the small intestine, arterial and venous endothelial cells and arterial smooth muscle cells in most organs. ACE2 mRNA expression is also found inthe cerebral cortex, striatum, hypothalamus, and brainstem.
The cerebral cortex, striatum, hypothalamus and brainstem are rich in
cholinergic neurons, and they are all part of our nervous system. It
also relates to our conscious self, learning and memory.
As I said at the beginning, a member of my close circle survived a
viral-induced fulminant myocarditis and a weird lung pathology. He was
out of the ICU and back at home before his fever recurred a couple of
weeks later and was treated successfully with piperacillin-tazobactam.
He doesn’t smoke either and he nearly died. He also received several
sessions of neural therapy (among many other therapeutic interventions)
to help him battle his cytokine storm. The good news is that, since
then, he has made an excellent recovery and is very pleased with the way
his brain is working. His thinking abilities are as good as they’ve
ever been. Not bad for someone who is over 75-years-old and does math
and physics for a living!
Further references
- Pavlov, Valentin A. The cholinergic anti-inflammatory pathway. Brain, Behavior, and Immunity 19 (2005) 493 – 499.
- Rosas-Ballina, M. et al. Cholinergic control of inflammation. Journal of Internal Medicine, 2009: 265; 663-679.
- Van Westerloo, David J., The vagal immune reflex: a blessing from above. Wiener Medizinische Wochenschrift (2010) 160/5 – 6:112 – 117.
APR



About the Author:
A countryside family medicine doctor and former heart surgeon in service of the Hippocratic Oath.